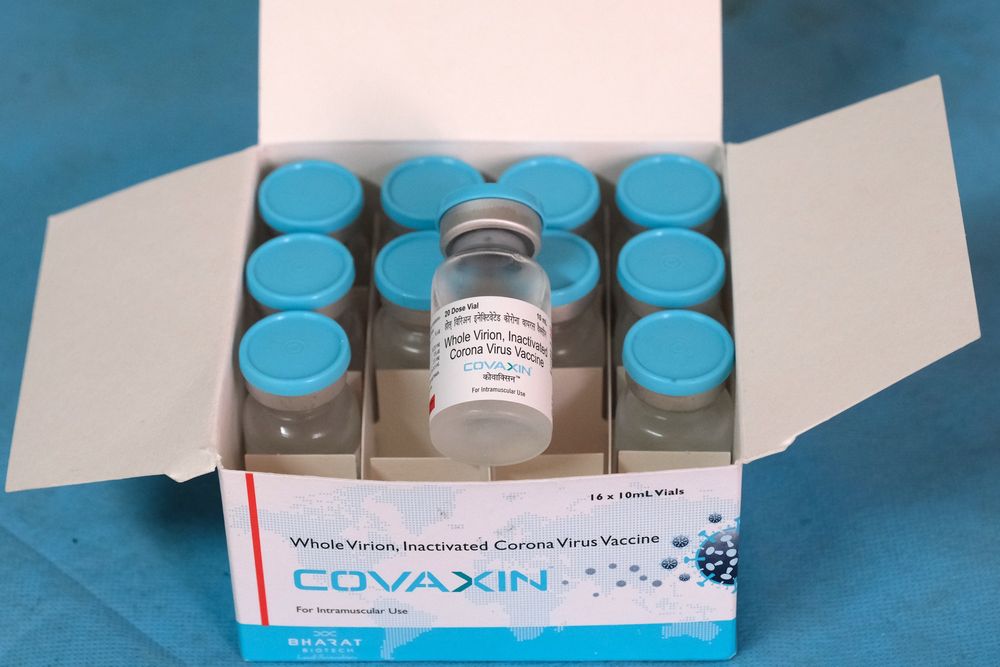
Covaxin, one of the COVID-19 vaccines in India, is not included in the World Health Organization’s emergency list.
Those who have been vaccinated with covaxin may not be allowed to travel internationally until the vaccine is approved. Only nine countries have approved Covaxin. At the same time, Covishield is recognized by 130 countries around the world.
When Bharat Biotech sent a recommendation on Covaxin to the World Health Organization, the World Health Organization guideline showed that “more information is needed”. Currently, about 20 million people in the country have received Covaxin. If approved by the World Health Organization, the vaccine can be imported from India to other countries that require the Covid-19 vaccination. According to the government, several countries have expressed interest in Covaxin.
The decision could affect international travel for those who have taken the covaxin vaccine, which has not yet been approved by the World Health Organization. Many countries, including Europe, want to resume international travel and open borders for international tourists. Most countries are expected to follow the World Health Organization’s EUL list when opening borders.
Covishield, meanwhile, is on the World Health Organization’s emergency list. Pfizer / Biontech vaccine, AstraZeneca / Oxford Covid-19 vaccine, Ad26.COV2.S developed by Johnson (Johnson & Johnson), and Moderna’s Covid-19 vaccine are approved by the World Health Organization.